What’s in Your Water?

Tap water is indisputably a more environmentally-friendly drinking choice than bottled water. But now, rising levels of pharmaceuticals in the water supply are raising concerns about the safety of the tap.
Whenever you take medication, not all of it is absorbed by your body – some passes right through you. Thousands of prescription and over-the-counter drugs are consumed in the U.S., as well as countless creams and ointments that are rubbed on and then showered off.
Traces of medications get flushed down the toilet and end up in our local water supply, which can endanger all tap water drinkers (for example, even small amounts of certain drugs can inhibit normal cell function and cause liver damage). Although our wastewater gets treated at sewage plants, it is difficult to remove every trace of drugs from the drinking water.
This situation has prompted four Chemical Engineering students at Ryerson University in Toronto, Canada to design a wastewater treatment system that could remove 90 percent of pharmaceuticals and other potentially harmful compounds.

Instead of chlorine, which most treatment plants use to disinfect the water, the students propose using ultraviolet light (UV) and hydrogen peroxide as an advanced way of oxidizing and disinfecting the water. The system, which was created using commercially available technology, would also use membrane biological reactors (see diagram below) to eliminate the murkiness of wastewater, which would then allow the UV light to penetrate and purify the water.
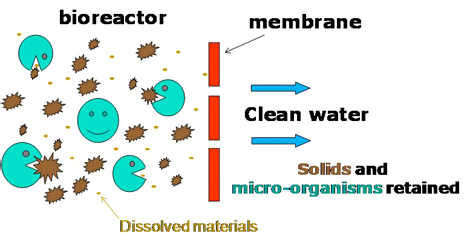
The project placed 1st for Social Awareness at the 2010 Ontario Engineering Competition and received an honorable mention for its innovative design. Now that’s pure genius!
Images:
Students – Dario Ruberto/Ryerson University
Diagram – Pierre Le Clech/Wikipedia








